A summer doing lab experiments as a doctoral student was enough for Yan Li. “I figured out I may be too clumsy to be a lab scientist” and eventually turned to computers, she says.
Li now is an assistant computational scientist at Brookhaven National Laboratory on New York’s Long Island, but that doesn’t mean she’s completely forsaken the lab bench. She often collaborates with experimental groups – partnerships that, among other things, study ways to make solar energy cheaper, more efficient and more flexible, whether to produce electricity or clean-burning hydrogen.
Most of Li’s work is motivated by what’s missing in experiments. “There are answers (scientists) can’t figure out simply by using experimental techniques or there are things they want to make predictions about before they try a new experiment. These are the areas theory, modeling and computation can help.”
Li focuses on first-principles electronic structure theory calculations: applying fundamental rules from the strange world of quantum mechanics while forsaking any adjustable parameters to compute the physical properties of materials, whether as molecules, surfaces, crystals or nanometer-sized structures.
The calculations are especially demanding because they adhere to quantum mechanical principles, which exact a higher computational cost than classical physics models. With first-principle electronic structure theory, Li and her colleagues decipher the detailed properties of materials at the atomic scale what happens at the interfaces where they meet.
Their favorite method: density functional theory (DFT), a generally accurate and reliable approach that takes much less computer time to reach a solution than quantum chemistry techniques. Its balance of accuracy and affordability makes DFT the most popular computational method for research in chemistry, materials science and other disciplines, Li says.
But “depending on the particular property you’re interested in, we have to go beyond DFT or combine” it with another method. That’s the case in one recent project to help solve a mystery involving gold nanoparticles, sunlight and hydrogen production.
Stony Brook University doctoral candidates Peichuan Shen and Shen Zao, with Alexander Orlov, materials science and engineering professor, study cadmium sulfide, a material that, when exposed to sunlight, mildly acts as a catalyst to produce hydrogen from water.
“It’s a standard substrate, not really fancy, but it has some unimpressive photocatalytic activity,” Li says. When the Stony Brook researchers seeded the cadmium sulfate with nanoparticles of different sizes and composition, they found gold clusters boosted hydrogen production by as much as 35 times.
The gold particles that worked best were about a nanometer in size – each just tens of atoms, as Brookhaven staff scientist Dong Su showed with high-tech transmission electron and scanning transmission electron microscopes. The researchers weren’t sure, however, what precise particle size contributed most to the enhanced effect or how they work. They could only average results from all the sizes on the substrate.
The Stony Brook researchers “have been scratching their heads trying to figure out what the mechanism is. … They were happy that it worked, and it works great, but they hadn’t figured out why and how. That’s where we came in.”
Li is working with graduate students on detailed theoretical studies to see what factors govern the enhanced hydrogen evolution: nanoparticle size or shape, chemical bonds with the substrate, or the modified electronic structure at the nanoparticle-substrate interface.
As a first step, Li used time-dependent DFT (TDDFT), a generalization of DFT, to calculate the nanoparticles’ light absorption properties.
TDDFT is necessary, Li says, because standard DFT can’t describe the excited electron structures. Like shifting a car into a higher gear, light kicks some electrons in the nanoparticles’ atoms into empty higher energy levels.
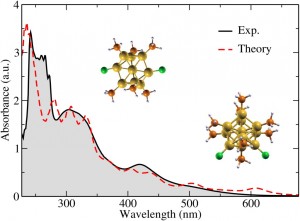
Using TDDFT, researchers can calculate the energy and strength of these optical transitions. The method lets them compare the nanoparticles’ computed ultraviolet-visible absorption spectra with spectra measured in experiments. Calculations also can reveal information not readily accessible from experiments, such as the nature of the electronic states involved in optical transitions.
The Stony Brook group, based on its analyses, suggested nanoparticles comprised of either nine or 11 gold atoms dominated the prepared samples. Li computed the corresponding structures and optical spectra of each and compared them with experimental results. “They agree quite well,” Li says. “That gives us some confidence that these are the dominating (particle sizes) in these experiments. The next stage is to look at how they interact with the substrate” to enhance hydrogen evolution so drastically.
Li and a graduate student are working on that problem with support from a Stony Brook seed grant. Eric Isaacs, a Department of Energy Computational Science Graduate Fellowship recipient from Columbia University, contributed to the project while on a practicum this summer.
Meanwhile, Li is looking at other, more complex nanostructures in collaboration with chemist Jonathan Owen and materials scientist Simon Billinge, both at Columbia. Owen’s group synthesized nanoparticles of cadmium-selenide, a material considered a good candidate for photovoltaics. They gauged the nanoparticles’ optical and infrared properties and, with help from BNL scientists, tracked how X-rays bounced off them. Billinge’s group then used data from the X-ray measurements to fit the nanoparticles’ structures.
However, such fitting procedures don’t consider the structures’ energetic stability, so the proposed solutions may not always be physically stable, Li says. And for complex nanostructures, fitting sometimes provides multiple solutions, each agreeing with X-ray measurements to a similar degree but providing few clues about which is the correct answer.
Li is performing DFT calculations to determine whether the proposed structures are physically real and stable. “We want to find out the exact structures of these nanoparticles, as well as how different structures and shapes influence optical properties,” Li says. Eventually, researchers could improve or modify the recipe for synthesizing particles to produce good candidates for photovoltaic materials.
But the models must account for ligands, molecules that attach to the nanoparticle surface, maintaining the surface’s semiconductor-like electronic structure. In the X-ray measurements, signals from the heavier elements cadmium and selenium overshadowed signals from the light atoms comprising the ligands, such as hydrogen, carbon, nitrogen and oxygen. As a result, the measurements tell researchers little about how the ligands bind to the nanoparticle surfaces.
By combining every datum the experiments collected with knowledge from scientific literature, Li plans to reduce the modeling configuration space and validate various stable structures she’s calculated with DFT.
The research tests Billinge’s complex modeling approach, which is based on the assertion that nanostructures’ complex nature makes them difficult to predict with a single technique – in this case, X-ray diffraction. Instead, it takes data from multiple experiments and techniques combined with theory to get at their true geometry.
So far, Owen and Li have identified the structure motif for the smallest cluster the Columbia group synthesized. Results are nearly ready for publication.
“It’s quite exciting,” Li adds. “If it works out we’ll have a methodology we can apply to similar, but bigger and more complex structures.”
It’s another example of the collaboration Li develops with scientists working at the lab bench. “It really takes frequent discussions to make sure we are consistent with each other – that what we compute is actually relevant and can help to understand and improve experiments.”
